Pest/Pathogen of the Month: October
Scientific name: Cercospora zeina Crous & U.Braun
Common names: Gray leaf spot
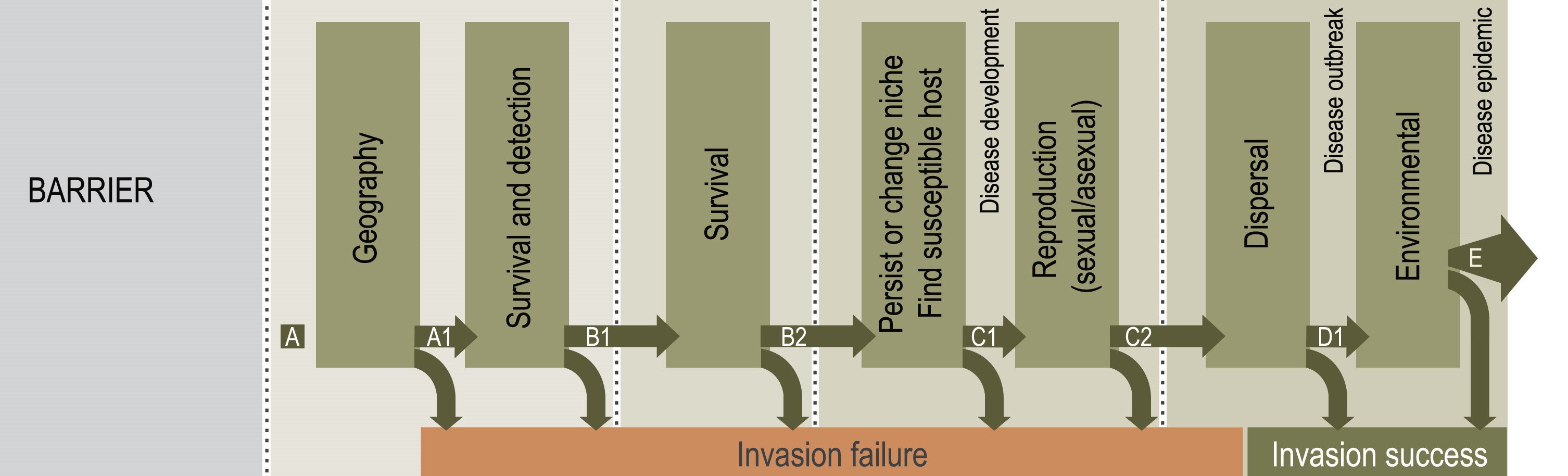
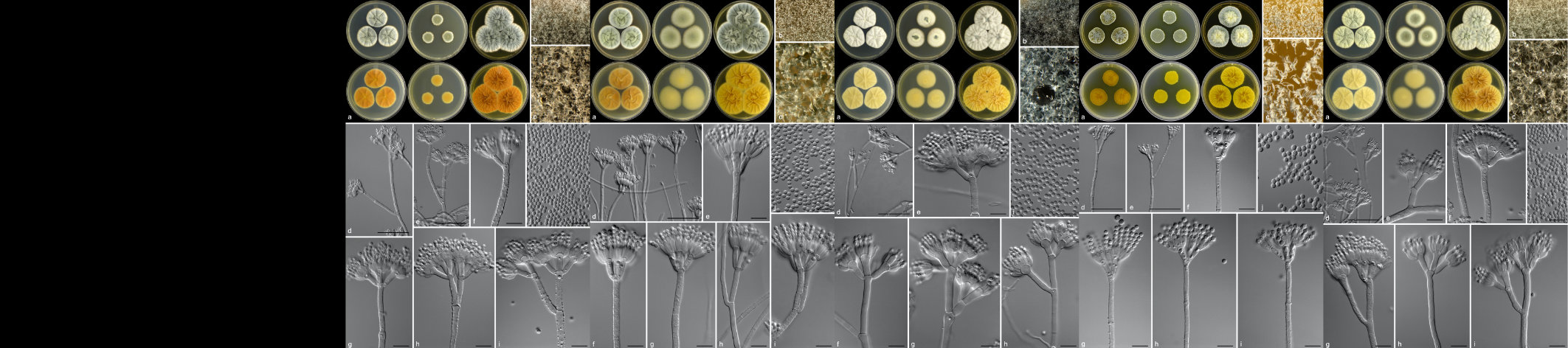
In Africa, Cercospora zeina is the predominant causal pathogen of gray leaf spot disease of maize. This fungus is a threat to food security, since it can cause severe yield losses on both small-holder and large-scale farms. The foliar symptoms caused by this fungus are tan to gray rectangular lesions that are restricted within veins of maize leaves. After consuming its “maize meal” inside the leaf, the fungus bursts out of the leaf stomata with a fresh set of spores to infect the next leaf. Many things still puzzle scientists about this fungus, such as the questions “Where did it come from?” and “How has it moved around Africa?” Gray leaf spot disease was first reported in the 1980’s in KwaZulu-Natal, South Africa. Reports of the disease have since emerged from other maize producing countries in sub-Saharan Africa. Could this be due to international and local trade of maize material that may contain infected leaf sheaths and ear husks, or has it been hiding in a wild grass species? Molecular analysis has shown C. zeina to have high genetic diversity in Africa and that it undergoes cryptic sex. No one has yet observed its sexual stage, but we suspect that this contributes to its high diversity and aggressiveness. Clearly, there is need to design and employ integrated pathogen management strategies to limit its reproduction and dispersal to ensure optimal maize production and food security in Africa and globally.

Pest/Pathogen of the Month: August
Scientific name: Sclerotinia sclerotiorum (Lib) de Bary.
Common names: white mold, cottony root, watery soft rot, stem rot, drop, crown rot and blossom blight.
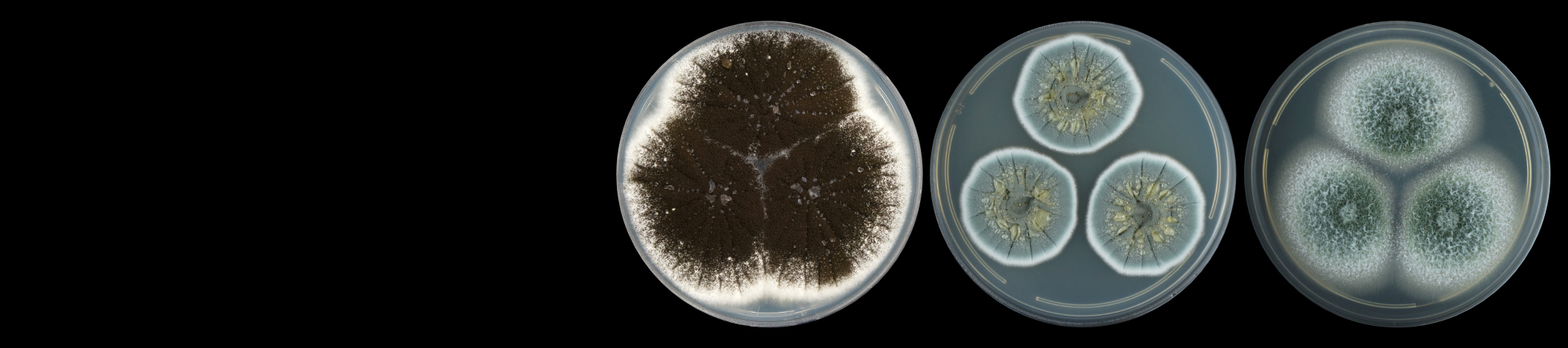
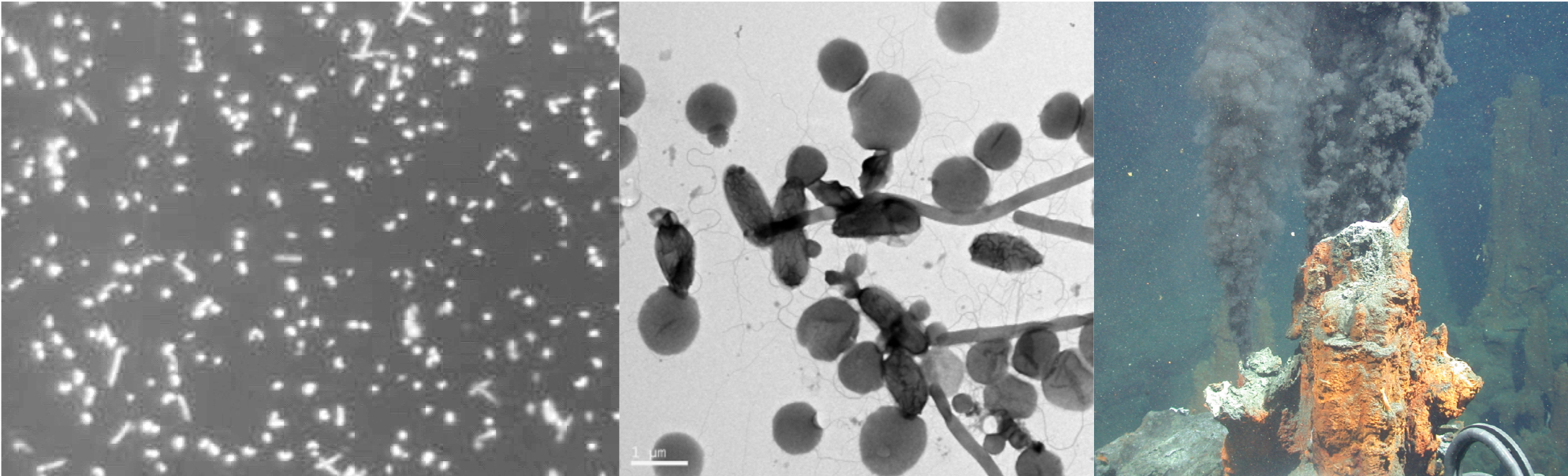
Sclerotinia sclerotiorum (Lib.) de Bary is an Ascomycete fungus in the order Helotiales. This species is a multi-host necrotroph that infects more than 400 plant species, with lettuce, sunflower, canola and sugar bean being of economic importance to the South African agriculture industry. Sclerotinia sclerotiorum is characterized by the production of long-term survival structures called sclerotia which form an important part of the infection cycle. These structures can lay dormant in the soil for up to 8 years until environmental conditions become conducive for germination. Carpogenic germination of sclerotia results in the formation of a sexual structure (the apothecia) at soil level, which releases millions of ascospores that start the infection cycle. Myceliogenic germination forms hyphae and mycelium that results in direct sub-terrain infection of host plants, although this is limited to a radius of about 2 cm around the sclerotia. Signs and symptoms of disease depend on the host plant infected and can include water-soaked lesions or dry lesions on stems, leaves, fruits, or petioles. The presence of white fluffy hyphae on the host surface during high humidity forms the basis of the name “white mold” that refers to the disease.
Photograph:Sclerotia (black structures) mixed in with soybean (Photo by Lisa Rothmann, taken from The South African Sclerotinia Research Network (SASRN) website)

Pest/Pathogen of the Month: September
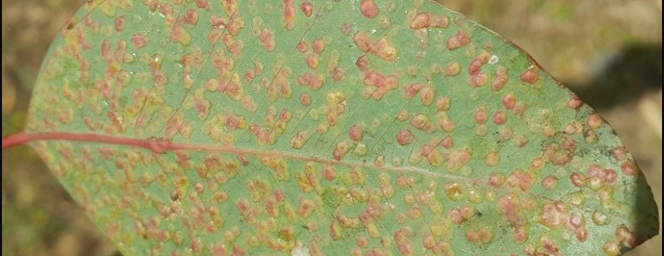
Scientific name: Spongospora subterranea f. sp.subterranea (Sss)
Common names: Powdery scab of potato
Powdery scab, caused by the obligate plant pathogen Spongospora subterranea f. sp. subterranea (Sss), is an unsightly blemish disease on potatoes and is a major problem in the potato industry worldwide. Powdery scab is identified by purple-brown pimple-like lesions that rupture the tuber periderm, creating powdery filled lesions. The powdery mass consists of masses of sporosori (collections of resting spores). These resting spores are highly resistant to unfavourable environmental conditions, allowing the pathogen to survive in the soil for over 50 years. The presence of these lesions reduces quality and marketability of seed tubers or tubers intended for consumption, causing major yield losses in potato production. This plant pathogen is also responsible for causing two other diseases, namely root infection and root galling, which also lead to yield reductions. Powdery scab disease is most severe in fields when the soil temperature is cool (9-17 °C) and has a high water content. Although infection occurs under cool and wet conditions, diseases have been recorded in hot and dry climates too, especially where irrigation is applied. The host range of Sss is broad as it infects plant species belonging to at least 26 families. Many weeds and commercial crop species have been confirmed to be alternative hosts of Sss. Powdery scab is difficult to successfully control due to the pathogen’s ability to form resting spores and the scarcity of resistant cultivars. No single control method can completely control Sss, however, an integrated management approach is advised for management of Sss.

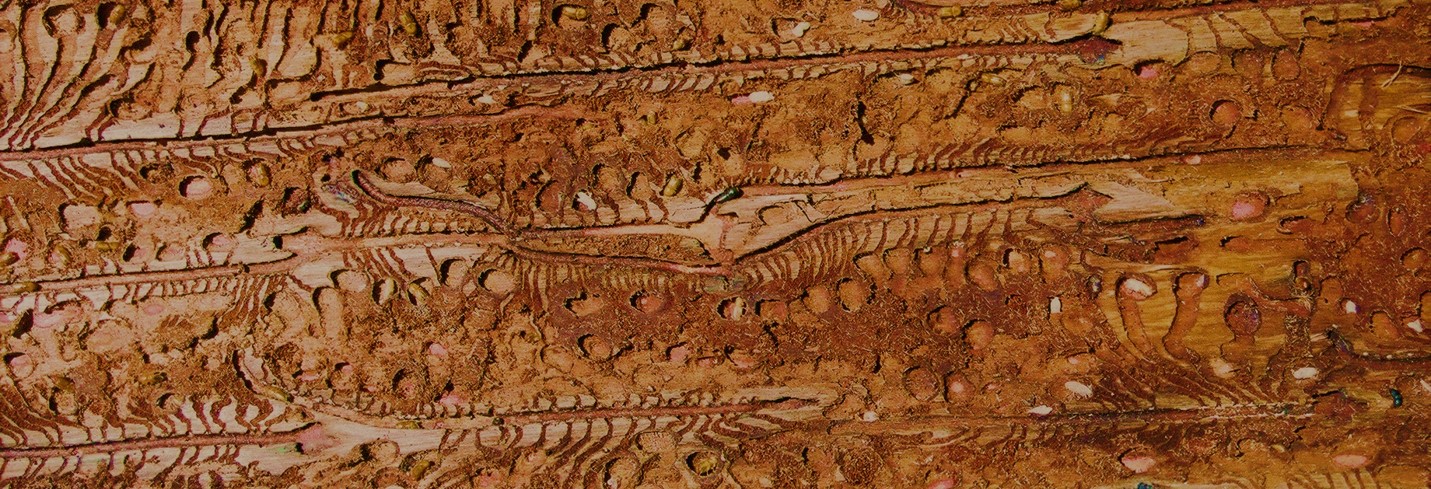
Pest/Pathogen of the Month: July
Scientific name: Teratosphaeria destructans
Common names:
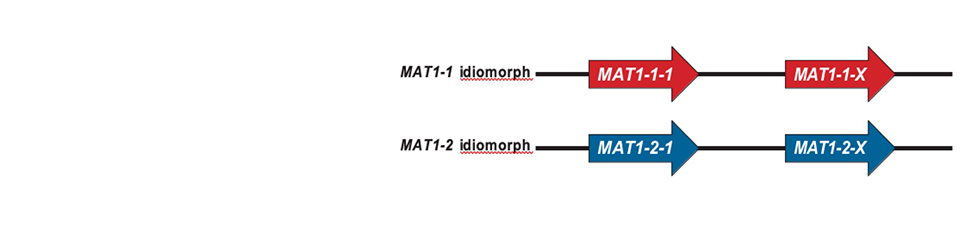
Teratosphaeria destructans is one of the most devastating pathogens to Eucalyptus forestry. It causes severe shoot and leaf blight on young Eucalyptus plantation trees and was, until recently, only known from South East Asia. In South Africa, T. destructans was first discovered in the KwaZulu-Natal Province by the FABI team in 2015. It has subsequently also spread to plantations of E. grandis and its hybrids in other sub-tropical parts of the country. Some good news is that the South African T. destructans population consists of a single genotype and mating type. This indicates that sexual reproduction is currently not possible and that the pathogen most likely entered South Africa as a single introduction. Care should be taken not to introduce the opposite mating type or additional genotypes.
Photo credit: https://www.fabinet.up.ac.za/index.php/news-item?id=274